You must be signed in to read the rest of this article.
Registration on CDEWorld is free. You may also login to CDEWorld with your DentalAegis.com account.
Lasers are an emerging technology in medicine, with many popular modalities, from laser eye surgery to laser hair removal, entering the mainstream. In dentistry, lasers provide better treatment options along with advanced technology, especially in regard to treatment of periodontal disease (PD). Diode lasers in particular can function as an adjunct to scaling and root planing (SRP). This article will evaluate the use of lasers in a hygiene protocol, including basic science topics as well as possible bactericidal effects and healing properties of soft-tissue diode lasers. Implementing lasers in the dental practice can lead to happier and healthier patients.
The Science and Technology Behind Lasers
The word laser is an acronym that stands for "light amplification by stimulated emission of radiation,"1 which describes the technology underlying this nonionizing tool. The history of lasers began with Einstein and his theory of stimulated emission. Einstein theorized that an atom in an excited state can absorb additional quanta of energy. Thus, when electrons return to a more stable state, they release two quanta of energy. This process of amplification and stimulated emission is the basis of creating laser light. Although Einstein never built a laser, his concepts were the foundation that led Theodore Maiman to fabricate the first working laser in 1960.1-3
The electromagnetic spectrum represents all forms of radiant energy, or wavelengths. Light waves, or "radiant wavelengths," are ubiquitous. Common units of measurement for wavelengths are µm and nanometer (nm). Visible light ranges from about 400 to about 750 nm. Wavelengths below 400 nm are considered ionizing radiation. Wavelengths above 750 nm are considered thermal radiation, or wavelengths that generate heat when absorbed by tissue. Diode lasers used in dentistry range from 810- to 1,064-nm wavelengths. These fall into the thermal range of the electromagnetic spectrum.3
Natural light is quite different from laser light. Natural light is a combination of multiple wavelengths of visible light emitted in different directions. The wavelengths are not aligned; therefore, natural light is considered incoherent and non-collimated. Because laser light is typically one wavelength, it is considered monochromatic. It is also coherent, meaning laser light waves travel in phase or alignment.4
A laser is made up of three parts. There is a pumping source, which provides energy to the laser system; an active medium, which is the material that is stimulated by the pumping source; and an optical chamber that contains the active media. This optical chamber is also referred to as a resonating chamber and is composed of two parallel mirrors. Laser light is created in the resonating chamber and delivered to the target tissue through a fiber optic.5
What Can a Soft-Tissue Diode Laser Do?
When laser energy comes into contact with soft tissue, a reaction occurs. Lasers have many uses in dentistry. They ablate, meaning they can incise, excise, or erase tissue. They coagulate or cauterize, sealing off blood vessels and creating a dry field or sealing off nerve endings. The latter function stops histamine release into the surgical site, lessening postoperative pain and inflammation. Lasers also decontaminate, or kill bacteria, which is the main focus of this article. Finally, they can biostimulate to promote healing. In dentistry, biostimulation is used for several procedures, including treatment of aphthous ulcers and herpetic lesions. Often, lasers can perform the aforementioned functions at the same time.6
A diode laser is also called a semiconductor laser because it has a semiconductor chip, or diode chip, that is doped or coated with active medium to create the desired wavelength. This transforms electrical energy into light energy. Lasers are named for their active medium, which can be a semiconductor, solid state, or a gas. For example, the active medium of a CO2 laser is a gas, carbon dioxide. Diode lasers range in wavelengths between 810 and 1,064 nm. The active medium for diode lasers generally includes a combination of gallium, arsenide, aluminum, and indium, depending on the wavelength.7
Because diode lasers have little to no effect on hard tissues, they are effective for multiple soft-tissue procedures and popular in dentistry. Surgically, diode lasers can effectively perform procedures such as gingivectomies and frenectomies.8 Because these lasers exhibit bactericidal and biostimulation properties, they are often used as an adjunct to periodontal procedures. Results of studies combining SRP with laser treatment have been favorable,9,10 making lasers an excellent choice for laser bacterial reduction and laser-assisted periodontal therapy.
However, there are differences in opinion concerning the efficacy of soft-tissue diode lasers as an adjunct to SRP. Although some studies show better results when lasers are used as an adjunct to SRP, others have demonstrated no statistically significant difference in an SRP-alone group compared with an SRP-plus-laser group.11,12 This variation in conclusions from differing studies can be explained by the multitude of variables considered in each, such as comparisons between which wavelength, setting, or technique was utilized.13,14 Personal experience and careful review of the literature can guide an appropriate treatment protocol and provide a sense of the efficacy and value of laser treatment.
A review including 1) the goals of SRP, 2) the need for adjunctive services based on bacterial load present in the mouth, and 3) the systemic and oral health impact of PD is necessary to determine the efficacy of diode lasers with SRP. Clinical studies supporting the role of diode lasers in periodontal therapy and their healing or treatment properties in the oral environment15,16 offer insight into the role of lasers in the dental practice.
Oral-Systemic Health
According to the Centers for Disease Control and Prevention, almost half the adult population older than 30 years has PD.17 A majority falls in the range of moderate to severe PD. The deleterious impact of chronic inflammation has become a large area of research. As more is understood about what initiates the inflammatory cascade and systemic effects of chronic inflammation, the medical profession has begun to look differently at conditions such as heart disease, diabetes, autoimmune disorders, and certain cancers.
Atherosclerosis has been connected to infectious, autoimmune, and inflammatory factors.18 In their review of studies connecting cardiovascular events and PD, Bale, Doneen, and Vigerust stated that PD caused by high-risk pathogens may be considered a contributory cause of arterial disease.19 They also postulated that the dental community has a substantial opportunity to mitigate the effects of this significant source of morbidity and mortality by elucidating feasible, effective management of PD.19
Dental professionals have several approaches to better manage chronic PD in their patients. Many of these methods can be used to more effectively treat PD, including salivary testing, chairside microscopes, systemic antibiotics, new antimicrobial products, and lasers. For patients diagnosed with PD, SRP is the gold standard of care and initial nonsurgical management of disease. The goals of nonsurgical periodontal therapy are: 1) eliminating diseased sulcular tissue, 2) minimizing intrasulcular bacterial activity, 3) halting apical migration of the epithelial attachment, 4) minimizing pocket depths, and 5) establishing a healthy oral environment the patient can easily maintain.20
However, SRP alone may not reduce the bacterial load present because tissue-invasive pathogens associated with PD reside in the subepithelial tissue.21 The pathogens are not necessarily destroyed by mechanical debridement performed during SRP.21 These gram-negative anaerobic bacteria are not only persistent but are also colonized in areas difficult to access with hand instruments. The following common bacteria are generally resistant to SRP: Actinobacillus actinomycetemcomitans, Porphyromonas gingivalis, Prevotella intermedia, and Peptostreptococcus micros.22-26 This is notable because bacteria from the mouth can enter the bloodstream.
According to Bale et al, the most common oral bacteria associated with atherosclerotic lesions are A. actinomycetemcomitans and P. gingivalis.19 Both are also associated with aggressive forms of PD. They are tissue-invasive and pathogenic, even at low bacterial counts. As the medical community becomes more familiar with the oral-systemic link and the role bacteria play in both chronic PD and overall health, clinicians should look beyond SRP alone and begin to consider lasers for PD treatment.27-29 Patient care should be developed from a comprehensive treatment plan that suits individual needs. The need to reduce bacterial count in the periodontal pocket is evident, making lasers as an adjunct to SRP an excellent treatment option.
Diode Lasers as Adjuncts to SRP
Over the last 20 years, multiple studies have evaluated the effectiveness of a diode laser's ability to reduce microbial colonies and improve clinical indices such as plaque index, gingival index, probing depths, and clinical attachment level.30-33 Consistently, these studies have shown a reduction in microbial count. In 1998, Moritz and a group of researchers published their findings on the treatment of periodontal pockets with a diode laser.34 They evaluated the laser's bactericidal abilities and examined bacterial count for A. actinomycetemcomitans, P. gingivalis, and P. intermedia.34 While evaluating periodontal conditions after the use of a diode laser, Moritz et al found that there was both a significant reduction in bacteria and improvement in clinical indices.34
A more recent study, Gupta et al in 2016, was intended to evaluate the effectiveness of diode lasers compared with SRP alone.30 The same clinical indices were measured in addition to obtaining a microbiologic sample for measurement of A. actinomycetemcomitans and P. intermedia bacterial loads.30 Patients participating in the study were reevaluated at 30, 90, and 180 days.30Gupta et al found that the group with SRP plus laser showed improvement in all areas and a higher reduction of A. actinomycetemcomitansand P. intermedia than the SRP-alone group.30
Of primary significance is that the SRP-plus-laser group saw better results and a long-term, continual decrease in bacteria.30 Gupta et al also commented on the bactericidal effects of diode lasers, stating that the wavelength of the diode laser is absorbed by pigmented anaerobic periopathogens.30 This leads to water vaporization and causes lysis (disintegration or rupture) of bacteria cell walls and creates bacterial cell gap. Additionally, although there are differing opinions on how laser energy interacts with tissues, the Gupta study and others35-37 have noted significant bactericidal effects of lasers.
In a 2015 systematic review, Qadri et al analyzed 43 studies on diode lasers in the treatment of chronic periodontitis and narrowed their examination down to 10 studies that met specific criteria.31 All 10 studies used diode lasers ranging from 808- to 980-nm wavelengths.31 Qadri et al set out to answer the question: Is SRP with diode-laser therapy more effective in the treatment of chronic PD than SRP alone?31 For this particular meta-analysis, all studies reviewed excluded patients with systemic conditions.31 In their findings, they determined that diode lasers operating at 808 to 980 nm with power between 0.8 and 2.5 watts provided good results as an adjunct therapy and confirmed the effectiveness of diode-laser therapy.31
Proper laser technique has been widely discussed, and there are differences of opinion on this topic. One variable is the type of tip that should be used for laser therapy as an adjunct to SRP. An initiated laser fiber is one that has been blackened on the end. This can be done by the clinician, but some laser manufacturers now provide initiated and noninitated tips. In general, a nonintiated tip is used for laser procedures between 50°C and 60°C. Therefore, a noninitiated tip is typically recommended for nonsurgical procedures, such as laser bacterial reduction and treatment of aphthous ulcers and herpetic lesions.38 For procedures performed at slightly higher temperatures, an initiated tip is usually recommended. An initiated tip will condense the laser energy to create a hot tip that is ideal for procedures to incise and excise tissue. An initiated tip will alter the outcome of the procedure, so it is important to become familiar with the correct settings, technique, and desired outcome. The clinician should also be familiar with the laser guidelines for an individual state and the scope of practice defined for dental hygiene. Detailed information on technique is covered in laser certification and training courses.
Healing Properties
Clinical studies have also identified the healing properties of diode lasers.39,40 They indicate an increase in cell proliferation,41 increased lymphatic flow, stimulation of immune response, and increased microcirculation. A 2012 study by Hakki and Bozkurt analyzed the effects of nonsurgical diode-laser applications using a 940-nm wavelength.42 They evaluated cell proliferation and mRNA expression of type I collagen in human gingival fibroblast growth factors.42 Hakki and Bozkurt compared three different laser settings and found that the lower biostimulation setting (0.3 watts in a continuous wave) was associated with significant collagen type I mRNA expression.42 They concluded that nonsurgical laser applications modulate the behavior of gingival fibroblasts and induce growth factors.42
Cell metabolism has been shown to increase due to biostimulation from a diode laser, thereby increasing the production of adenosine triphosphate, the fuel that powers cells.30 This increase in energy is available to normalize cell function and promote tissue healing.30 Biostimulation can be defined as a laser's ability to promote healing. The healing effects of lasers are noted in multiple studies.43,44
Tissue Interaction Properties
When using a laser, multiple variables can determine the effect on biologic tissue. Wavelengths react on target tissue in three different ways: absorption, reflection, and transmission. For absorption, chromophores absorb light energy, which is then converted to heat to perform work. This is primarily how diodes function. Reflection occurs when a laser beam bounces off tissue and there is no interaction. Transmission is when laser energy passes through to deeper tissues. This is seen with diode lasers. "Scatter" occurs when the laser energy goes in different directions, which has been shown to produce biostimulative properties.45
Variables affecting laser-tissue interaction include wavelength, target composition, interaction time, energy transfer mode, fiber diameter/spot size, and operator knowledge, experience, and technique.3 When working with diode lasers, it is first important to consider that different wavelengths interact differently with tissue.
In considering target composition, chromophores such as water and pigments such as hemoglobin and melanin are light absorbers. Most diodes function through absorption, so depending on tissue combination, laser energy may react differently. This is exemplified by the difference in response between patients with highly inflamed tissue and those with thick fibrotic tissue.3 Finally, interaction time, whether the laser is in contact with the tissue or used in a noncontact mode, and fiber diameter size, in addition to clinician knowledge, will impact the success of laser use.
Case Example: Diode Lasers for Healing
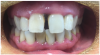
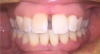
Figure 1 and Figure 2 show the preoperative and postoperative images of a 30-year-old patient who had not had a dental checkup in a few years. She was going to a wedding, which prompted her to visit the dentist for whitening treatment. This patient was completely unaware she had PD. She was treated with SRP, as well as laser therapy. Figure 2 shows the postoperative image taken on the same day that the left-side SRPs were completed. The patient responded well to treatment and was happy with the results.
Getting Started With Lasers
Several types of lasers on the market offer specific advantages to dental professionals. This section will cover tips for choosing a laser, research necessary to determine the optimal laser for unique needs, and where to find proper training to achieve success. It will also cover state guidelines, safety, and teamwork tips to develop a hygiene protocol that includes diode lasers.
Choosing a Laser
Several factors must be considered when selecting a laser.46 First, what procedures does the clinician intend for the laser's use? If the clinician knows he or she wants to use the laser as an adjunct to SRP, the clinician may also want to consider other procedures currently practiced in the office that could be done with a laser. Wavelength is also an important consideration. Will the wavelength be effective in performing the desired procedures? Services not currently performed in the practice could also be considered.
After determining the procedures for the laser to perform, finding a laser that meets related needs may be challenging. Here are some tips on how to begin researching for an ideal laser:
•
Take a laser course and get trained. Know the science behind lasers. Based on the wavelength, each laser will have specific clinical indications. Taking a course before purchasing a laser will help to determine the best wavelength.
•
Visit the Academy of Laser Dentistry (ALD)'s website, laserdentistry.org. It contains a significant amount of information, including articles, blogs, news, and publications on lasers. Going through the ALD first provides unbiased information from industry leaders with clinical experience.
•
Reach out to peers through dental networking groups to get feedback and reviews.
•
After the decision has been narrowed to a few choices, contact the manufacturers of the lasers for demonstrations. "Test drive" the laser before purchase. Laser devices from several different manufacturers have received US Food and Drug Administration clearance for periodontal procedures to improve clinical indices.
It is essential to sign up for a laser training course. Then, after a specific laser is chosen, the manufacturer should be contacted to discern what type of individual or team training is available. It is important for even nonclinical team members to understand the safety parameters of a laser.
State Guidelines and Safety Standards
State regulatory guidelines for laser training requirements should be verified. Rules may vary significantly from state to state. Additionally, some states require laser registration with the state radiation regulatory office. Clinicians should ensure they are working within the scope of practice specific to their state, which may prevent problematic situations in the future.
The American National Standards Institute (ANSI) defines the requirements for safe and effective use of lasers in healthcare. ANSI guidelines and requirements are typically covered in a laser training course. Some states have specific regulatory agencies that oversee radiation safety, in which lasers are included. Clinicians should check individual state requirements for additional safety protocols.
Team Approach
Implementing lasers as an adjunct to SRP necessitates a team approach that begins with training the entire staff. Clinicians will need specific training that meets ANSI required standard proficiencies as well as individual state curriculum guidelines to stay within their scope of practice. Nonclinical staff should be trained in laser safety to better maintain a safe environment for themselves and patients. All clinicians in the practice will need a thorough understanding of the role a diode laser plays in periodontal therapy and as an SRP adjunct. This ensures the purpose of laser use can be effectively communicated to patients. The team must decide where a laser fits into the office's hygiene protocol.
The Hygiene Appointment
Successfully integrating lasers into a dental practice's periodontal protocol begins with a solid explanation and frame-up of PD. The patient must understand the benefits of periodontal therapy and adjunctive services. Fortunately, patients frequently hear about lasers in medicine. Studies show that the majority of patients believe lasers will make their visit to the dentist easier,47,48 but they will have questions about the laser.
The most common patient questions regarding lasers include the following:
• Does the laser hurt? When using the laser for SRP procedures, whatever anesthetic is used for the SRP will be sufficient for the laser. Often, patients will not need anesthetic for laser therapy. Generally, using a laser for periodontal maintenance appointments will not require anesthetic. However, if a patient is concerned about sensitivity, the author has found that a topical anesthetic works well.
• How much will it cost? When lasers are used as an SRP adjunct, cost can vary significantly. It is best for a clinician to devise the fee based on the customary fees charged in the particular geographic location or practice style.
• Will insurance cover the procedure? Often, insurance does not cover laser use as an SRP adjunct. Some insurance plans consider laser therapy an inclusive part of the SRP procedure. For those who must pay for the additional service, the cost tends to be affordable.
Conclusion
Many clinicians will benefit from using lasers in their hygiene protocol, which can be an intellectually stimulating and productivity-boosting addition to the dental practice. Diode lasers also provide oral health benefits to patients. By understanding the science and technology behind lasers, in addition to important factors such as the oral-systemic impact of PD, laser healing properties, and tissue interaction, clinicians can effectively incorporate laser use into their office procedures. This service requires a team approach, careful product selection, thorough training, and excellent patient communication.
References
1. Ritter AV. Lasers in dentistry. J Esthet Restor Dent. 2006;18(1):58.
2. This month in physics history. APS News. 2005;14(8). https://www.aps.org/publications/apsnews/200508/history.cfm. Accessed December 19, 2018.
3. Coluzzi DJ. An overview of laser wavelengths used in dentistry. Dent Clin North Am. 2000;44(4):753-765.
4. Romanos GE. The state of the science of lasers in dentistry. J Dent Hyg. 2012;86(1):9-10.
5. Svelto O, Hanna DC. Principles of Lasers. New York, NY: Plenum Press; 1998.
6. Pang P, Andreana S, Aoki A, et al. Laser energy in oral soft tissue applications. J Laser Dent. 2010;18(3):123-131.
7. Dowst-Mayo L. Dental hygiene lasers: why you should use lasers. RDH. 2012;30(10):54-60.
8. Pick RM, Colvard MD. Current status of lasers in soft tissue dental surgery. J Periodontol. 1993;64(7):589-602.
9. Giannelli M, Materassi F, Lorenzini L, Bani D. Management of severe periodontal abscesses using a laser and light-emitting diode procedure adjunctive to scaling and root planing: a case series. Clin Adv Periodont. 2017;7(3):159-166.
10. Crispino A, Figliuzzi MM, Iovane C, et al. Effectiveness of a diode laser in addition to non-surgical periodontal therapy: study of intervention. Ann Stomatol (Roma). 2015;6(1):15-20.
11. Cobb C. Lasers in periodontics: a review of the literature. J Periodontol. 2006;77(4):545-564.
12. Schwarz F, Sculean A, Berakdar M, et al. In vivo and in vitro effects of an Er:YAG laser, a GaAlAs diode laser, and scaling and root planing on periodontally diseased root surfaces: a comparative histologic study. Lasers Surg Med. 2003;32(5):359-366.
13. Takasaki AA, Aoki A, Mizutani K, et al. Application of antimicrobial photodynamic therapy in periodontal and peri-implant diseases. Periodontol 2000. 2009;51(1):109-140.
14. Porteous MS, Rowe DJ. Adjunctive use of the diode laser in non-surgical periodontal therapy: exploring the controversy. J Dent Hyg. 2014;88(2):78-86.
15. Tewari M, Rai P, Singh GB, et al. Long-term follow-up results of Nd: YAG laser treatment of premalignant and malignant (stage 1) squamous cell carcinoma of the oral cavity. J Surg Oncol. 2007;95(4):281-285.
16.Aoki A, Mizutani K, Schwarz F, et al. Periodontal and peri-implant wound healing following laser therapy. Periodontol 2000. 2015;68(1):217-269.
17. Eke PI, Dye BA, Wei L, et al; CDC Periodontal Disease Surveillance workgroup. Prevalence of periodontitis in adults in the United States: 2009 and 2010. J Dent Res. 2012;91(10):914-920.
18. Sherer Y, Shoenfeld Y. Atherosclerosis. Ann Rheum Dis.2002;61(2):97-99.
19. Bale BF, Doneen AL, Vigerust DJ. High-risk periodontal pathogens contribute to the pathogenesis of atherosclerosis. Postgrad Med J. 2017;93(1098):215-220.
20. Sweeting LA, Davis K, Cobb CM. Periodontal Treatment Protocol (PTP) for the general dental practice. J Dent Hyg.2008;82(suppl 3):16-26.
21. Slots J, Ting M. Systemic antibiotics in the treatment of periodontal disease. Periodontol 2000. 2002;28(1):106-176.
22. Mombelli A, Gmür R, Gobbi C, Lang NP. Actinobacillus actinomycetemcomitans in adult periodontitis. II. Characterization of isolated strains and effect of mechanical periodontal treatment. J Periodontol. 1994;65(9):827-834.
23. Jain S, Darveau RP. Contribution of Porphyromonas gingivalis lipopolysachharide to periodontitis. Periodontol 2000. 2010;54(1):53-70.
24. Wadhwani RB, Chaudhary MS, Tharani DA, Chandak SA. Effect of scaling and root planing on detection of Tannerella forsythia in chronic periodontitis. J Oral Dis. 2013. doi: 10.1155/2013/383746.
25. Predin T, Djuric M, Nikolic N, et al. Clinical and microbiological effects of quadrant versus full-mouth root planing-a randomized study. J Dent Sci. 2014;9(4):400-406.
26. Everett JD, Rossmann JA, Kerns DG, Al-Hashimi I. Laser assisted non-surgical periodontal therapy: a double blind, randomized clinical trial. Open Dent J. 2017;11:79-90.
27. Aoki A, Sasaki K, Watanabe H, Ishikawa I. Lasers in nonsurgical periodontal therapy. Periodontol 2000. 2004;36(1):59-97.
28. Ishikawa I, Aoki A, Takasaki A. Potential applications of erbium:YAG laser in periodontics. J Periodontal Res.2004;39(4):275-285.
29. Ishikawa I, Aoki A, Takasaki AA, et al. Application of lasers in periodontics: true innovation or myth? Periodontol 2000. 2009;50(1):90-126.
30. Gupta S, Sawhney A, Gourav J, et al. An evaluation of diode laser as an adjunct to scaling and root planing in the nonsurgical treatment of chronic periodontitis: a clinico-microbiological study. Dent Med Res. 2016;4(2):44-49.
31. Qadri T, Javed F, Johannsen G, Gustafsson A. Role of diode lasers (800-980 nm) as adjuncts to scaling and root planing in the treatment of chronic periodontitis: a systematic review. Photomed Laser Surg. 2015;33(11):568-575.
32. Birang R, Yaghini J, Adibrad M, et al. The effects of diode laser (980 nm wavelength) and chlorhexidin gel in the treatment of chronic periodontitis. J Lasers Med Sci. 2011;2(4):131-138.
33. Bozkurt SB, Hakki EE, Kayis SA, et al. Biostimulation with diode laser positively regulates cementoblast functions, in vitro. Lasers Med Sci. 2017;32(4):911-919.
34. Moritz A, Schoop U, Goharkhay K, et al. Treatment of periodontal pockets with a diode laser. Lasers Surg Med. 1998;22(5):302-311.
35. Jawhara S, Mordon S. Monitoring of bactericidal action of laser by in vivo imaging of bioluminescent E. coli in a cutaneous wound infection. Lasers Med Sci. 2006;21(3):153-159.
36. Ando Y, Aoki A, Watanabe H, Ishikawa I. Bactericidal effect of erbium YAG laser on periodontopathic bacteria. Lasers Surg Med. 1996;19(2):190-200.
37. Cobb CM, McCawley TK, Killoy W. A preliminary study on the effects of the Nd:YAG laser on root surfaces and subgingival microflora in vivo. J Periodontol.1992;63(8):701-707.
38. Gemini 810 + 980 diode laser user manual. Ultradent. https://intl.ultradent.com/es-la/Product%20Instruction%20Documents/Gemini.pdf. Accessed March 1, 2019.
39. Roodenburg JLN, Panders AK, Vermey A. Carbon dioxide laser surgery of oral leukoplakia. Oral Surg Oral Med Oral Pathol. 1991;71(6):670-674.
40. López-Jornet P, Camacho-Alonso F. Comparison of pain and swelling after removal of oral leukoplakia with CO2 laser and cold knife: a randomized clinical trial. Med Oral Patol Oral Cir Bucal. 2013;18(1):e38-e44.
41. Soares D, Ginani F, Henriques ÁG, Barboza C. Effects of laser therapy on the proliferation of human periodontal ligament stem cells. Lasers Med Sci. 2015;30(3):1171-1174.
42. Hakki SS, Bozkurt SB. Effects of different setting of diode laser on the mRNA expression of growth factors and type I collagen of human gingival fibroblasts. Lasers Med Sci. 2012;27(2):325-331.
43. Lee JY, Lee SU, Lim T, Choi SH. Healing effects and superoxide dismutase activity of diode/Ga-As lasers in a rabbit model of osteoarthritis. In Vivo. 2014;28(6):1101-1106.
44. Yadav A, Gupta A, Keshri GK, et al. Photobiomodulatory effects of superpulsed 904nm laser therapy on bioenergetics status in burn wound healing. J Photochem Photobiol B. 2016;162:77-85.
45. Pohlhaus SR. Lasers in dentistry: minimally invasive instruments for the modern practice. Tissue interactions and biological effects. Procter & Gamble. https://www.dentalcare.com/en-us/professional-education/ce-courses/ce394/tissue-interactions-and-biological-effects. Accessed January 18, 2019.
46. A comprehensive buying guide to dental lasers. Academy of Laser Dentistry. http://www.laserdentistry.org/uploads/files/misc/Laser_Buying_Guide.pdf. Accessed December 19, 2018.
47. Wigdor H. Patients' perception of lasers in dentistry. Lasers Surg Med. 1997;20(1):47-50.
48. Zhegova GG, Rashkova MR, Yordanov BI. Perception of Er-YAG laser dental caries treatment in adolescents - a clinical evaluation. J IMAB. 2014;20(1):500-503.